The release of the FY2027 SNF PPS proposed rule, CMS-1843-P, brings several meaningful shifts that will impact how we approach documentation

As an MDS Coordinator, regulatory updates are not just policy changes. They directly shape our daily workflows, interdisciplinary collaboration, and ultimately, the story our data tells about resident care. The release of the FY 2027 SNF PPS proposed rule, CMS-1843-P, brings several meaningful shifts.that will impact how we approach documentation, submission timelines, and quality reporting.
From Retrospective to Real-Time: The Push for Faster Data
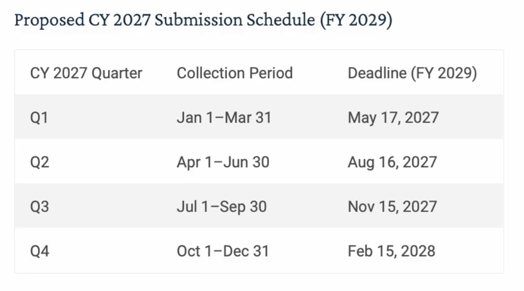
One of the most notable proposals is the shortened submission timeframe under the SNF Quality Reporting Program (QRP), moving from approximately 4.5 months after the end of each quarter to the 15th day of the second month after the end of the quarter, or by 11:59 p.m. of the following business day if the 15th falls on a Friday, weekend, or federal holiday, beginning with FY 2029 SNF QRP. CMS is also proposing a related change to the SNF Value-Based Purchasing Program snapshot date to align with that timeline.
All-Payer MDS Submission: Expanding the Scope
The proposal would require MDS data submission for all residents receiving skilled services regardless of payer source, using the Nursing Home PPS (NP) and the Nursing Home Part A PPS
Discharge (NPE) assessments, beginning with residents admitted for covered skilled services on or after October 1, 2029. With this change, the MDS evolves from a Medicare-centered reporting tool to a more comprehensive system supporting facility-wide clinical and quality initiatives.
Potential benefits include:
- A more accurate representation of SNF quality of care
- More standardized data across payer types and better support for data exchange between SNFs and other providers
- Stronger care coordination, continuity in care planning, and discharge planning
- Better healthcare outcomes for beneficiaries
At the same time, it would increase assessment volume, add tracking complexity, and require tighter processes across Medicare, managed care, and Medicaid populations.
The proposed rule would also include a modification to one MDS item in the Type of Assessment section to indicate when an assessment is being completed at admission for a non-Medicare FFS resident receiving skilled services, along with three new MDS items:
- Primary payer at admission and discharge from covered skilled services
- Start and end date of the covered skilled stay for a non-Medicare FFS resident
- A new Type of Assessment item to indicate whether the assessment is being completed for a non-Medicare FFS resident at the time of discharge from covered skilled services
Saying Goodbye to COVID-Era Measures Is a Welcome Trend
CMS has proposed removing two COVID-19 vaccination measures beginning with FY 2028 SNF QRP: the COVID-19 Vaccination Coverage Among Healthcare Personnel Measure and the COVID-19 Vaccine: Percent of Patients/Residents Who Are Up to Date Measure. While this reflects an evolution beyond the pandemic, it also highlights a broader trend: quality measures are dynamic and responsive to current healthcare priorities.
Increased Scrutiny of PDPM and Coding Accuracy
The proposed rule also includes a Request for Information on methodology for quantifying and addressing what CMS describes as case-mix creep under PDPM. CMS does not present this section as an adopted FY 2027 payment cut. At the same time, this is more than a casual policy discussion. CMS says it is “exploring a potential approach,” outlines a methodology, shows what that methodology could produce, and states that the approach it is considering “could be included in future rulemaking.”
CMS points to notable increases in some payment-related MDS items since PDPM implementation, including malnutrition, swallowing disorders, and depression. In the technical methodology memo accompanying the rule, CMS says reporting of the malnutrition item increased from 5 percent of stays in FY 2018 to 47 percent in FY 2024, swallowing disorder increased from 4 percent to 21 percent, and depression increased from 4 percent to 19 percent. CMS is using these trends to support its concern that some case-mix growth may reflect coding or documentation intensity rather than true changes in resident acuity or resource use.
CMS included illustrative adjustment factors under the methodology it is presenting for comment. Under that example, the factors would be +3.3 percent for PT, +4.1 percent for OT, -15.9 percent for SLP, -1.9 percent for NTA, and -10.6 percent for Nursing. CMS also showed an alternative system-wide factor of 0.957, which it says can be interpreted as a 4.3 percent reduction in case-mix adjusted payment, or approximately a 3.6 percent reduction in total payment across the system once the non-case-mix portion is included.
Even modest percentage changes can translate into meaningful losses for a high-volume Medicare Part A facility. Using CMS’s own illustrative rate example and applying all of the component-level factors CMS included for comment, the total PDPM per diem would fall from $1,139.87 to about $1,104.09, a reduction of roughly 3.1 percent overall. For a facility with about $10 million in annual Medicare Part A revenue, that would translate to an estimated loss of about $314,000 per year. Under CMS’s broader alternative system-wide approach of approximately 3.6 percent, the impact on that same revenue base would be about $360,000 annually. Facilities with more modest Medicare Part A revenue can apply the same math to estimate their own potential exposure. These figures are illustrative, not finalized policy, but they show why this issue deserves close attention.
From the MDS Coordinator’s chair, this reinforces the importance of accurate, defensible coding, strong collaboration with therapy, nursing, direct care staff, and providers, and ongoing internal auditing and education. This is not simply about compliance. It is about making sure the data accurately reflects resident acuity and the care actually being provided.
What This Means for the IDT
Perhaps the most important takeaway is that these changes cannot be owned by the MDS department alone.
Success under these proposals will require:
- Stronger interdisciplinary communication
- Real-time documentation from nursing and ancillary departments
- A shared understanding that data equals reimbursement, quality scores, and public reporting
The role of the MDS Coordinator continues to expand beyond assessment completion alone. It increasingly includes managing the flow of information across the interdisciplinary team and helping ensure that documentation supports both clinical accuracy and regulatory compliance.
Comments on CMS-1843-P are due June 1, 2026.
Staying ahead of regulatory changes like the FY 2027 proposed rule is not just about compliance. It is about making sure documentation processes, interdisciplinary communication, and internal oversight are strong enough to support quality care and accurate reporting.
Polaris Group can help facilities evaluate current MDS workflows, strengthen documentation practices, and prepare operationally for what may be ahead.

